- November 7, 2019
- Category: Press release
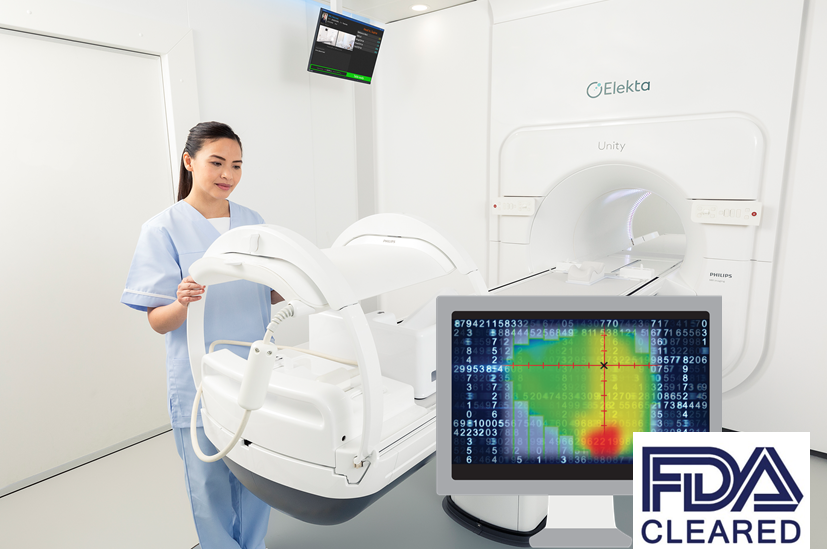
Cachan| Thursday, November 7th, 2019
DOSIsoft, leading provider of patient-specific software solutions for Radiation Oncology and Nuclear Medicine, is pleased to announce that on October 24th, 2019, it received 510(k) clearance from the US Food and Drug Administration (FDA), to market MU2net, software dedicated to Independent Secondary Dose Calculation for Patient Quality Assurance (QA).
MU2net: Providing a Fast TPS Plan Verification for Flexible Patient QA
Fast, automated and reliable, MU2net is a quality control tool for external beam Radiation Therapy that independently verifies the dose calculation performed by the Treatment Planning System (TPS) to check for potential errors in the patient treatment plans. It is an important part of Patient-Specific Quality Assurance (QA) for radiotherapy, together with pre-treatment beam verification and in vivo dosimetry.
MU2net is vendor neutral and compatible with all current treatment techniques, making it the ideal solution for every clinic, irrespective of existing infra-structure . The latest release of MU2net (version 2.1.1) also extends its compatibility to the Elekta MR-Linac, Unity.
By providing results within seconds, MU2net is perfectly positioned to support new adaptive workflows and enables the user to quickly react to changes while at the same time minimizing the workload. This is essential for the latest standards of personalized adaptive radiation treatment where plans are adapted to account for daily changes in shape, size and position of both the tumor and surrounding healthy anatomy. This guarantees safe dose delivery.
MU2net: Bringing a Cost-effective Secondary Dose Calculation Solution to the US Market
MU2net has been designed to offer a cost-effective solution for RT clinical routines. Already used clinically in Europe for several years, its newly obtained 510(k) FDA clearance allows MU2net to be marketed in the US and enables US RT departments to perform TPS plan check in an easy and affordable way. Accessing MU2net solution will also help the US RT departments to meet the current QA regulatory compliance.
MU2net is the latest Patient QA solution from DOSIsoft to receive 510(k) FDA clearance. With EPIbeam for pretreatment beam verification and EPIgray® for in vivo dosimetry, it completes the full DOSIsoft Patient QA portfolio to provide a verification at each step of the treatment course. Following the recent exclusive distribution agreement with Elekta, this Patient QA suite is available as part of the Elekta Assurance QA portfolio.
“The recent FDA clearance is an important milestone for the development of DOSIsoft in the US market. MU2net has been used in many European Radiation Therapy centers for several years as our team built a great solution to answer the clinical needs, using an algorithm independent from their TPS to run a fast calculation and providing results in a very user-friendly interface accessible via web from any device connected to their network. With the newer RT techniques available, patient-specific QA solutions have become even more important. Our team is very proud to launch in the US market this secondary dose calculation compatible with the Elekta MR-Linac Unity, and we look forward to deploying it in collaboration with our partner Elekta.” said Jean-Elie KAFROUNI, CEO of DOSIsoft Inc.
About DOSIsoft
Founded in 2002, DOSIsoft develops patient-specific software solutions for Radiation Oncology and Nuclear Medicine. 17 years of innovation and R&D investments have led to world leading software solutions used successfully in over 200 hospital centers in 26 countries around the world. Spin-off between Gustave Roussy and Institut Curie, DOSIsoft constantly innovates in partnership with the major cancer institutes and research centers in the world. www.dosisoft.com
“Image courtesy of Elekta” for Elekta MR-Linac, Unity illustration
