- October 12, 2018
- Category: Press release
Cachan| Friday, October 12th, 2018
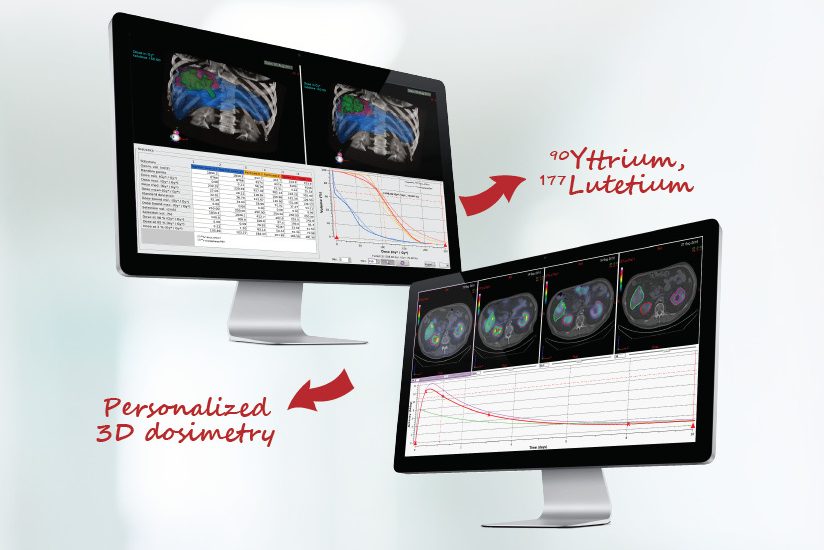
DOSIsoft SA, leading provider of advanced software solutions for Radiation Oncology and Nuclear Medicine, launches a new version of PLANET® Dose – its multi-radionuclide dosimetry platform dedicated to Molecular Radiotherapy (MRT). In addition to improvements for 90Yttrium-based therapies, the new version includes the full support of 177Lutetium-based therapies. These additional developments have been performed in collaboration with the Cancer Research Center of Toulouse (CRCT) – UMR 1037 Inserm / Toulouse III – Paul Sabatier University and the Institut Jules Bordet in Brussels. PLANET® Dose is the first CE marked platform on the market supporting 3D personalized dosimetry for 90Yttrium and 177Lutetium therapies.
PLANET® Dose V3.1.1: multi-radionuclide 3D voxel-based dosimetry platform
Complete, versatile, high performance and vendor-neutral treatment planning software solution, PLANET® Dose offers a multi-radionuclide platform supporting 90Yttrium/SIRT therapies and 177Lutetium-based radionuclide therapies. This new version enhances the previous release with specific functionalities for systemic therapies, in particular related to the pharmacokinetic modelling required for such treatments. The advanced features include: multi-time point quantification, elastic image registration, automatic deformable propagation of Volume-Of-Interest across times, residence time calculation, dose computation, consolidation of multi-treatment stages as well as advanced dose distribution analysis.
Personalized 3D dosimetry for Lutetium-based therapies
177Lutetium-based therapies are emerging and promising forms of Peptide Receptor Radionuclide therapies (PRRT), targeting tumors with radiolabeled molecules that bind to specific receptors expressed by the tumors. Main indications of 177Lutetium-based therapies are neuroendocrine tumors (NETs).
Compared to 90Yttrium/SIRT therapies, 177Lutetium-based therapies belong to the family of systemic therapies, for which the modelling of the pharmacokinetics of administered radiopharmaceutic is key to compute an accurate 177Lutetium absorbed dose deposition in different parts of the body, organs at risk (e.g. kidneys) and tumor sites.
Today, 177Lutetium-based therapies are mainly performed with standard protocols based on fixed activities and intervals between treatment cycles. Like other types of ionizing radiation therapies (External Beam Radiation Therapy huge progresses over the last ten years have demonstrated this) the personalized dose computation, with the double objective to (1) increase the absorbed dose to the tumor, and (2) maintain acceptable dose levels at organs at risk / healthy tissues, has the potential to significantly improve the therapeutic efficiency while mastering the patient safety and treatment tolerance. An individualized dosimetry-based approach will enable the delivery of higher radioactive activities at tumor sites while preserving in particular renal functions and other healthy organs.
PLANET® Dose meets the new European regulatory requirements (2013/59 Euratom)
The need of doing verification of the absorbed dose delivered to individual patient and the importance of performing personalized, dosimetry-based treatment planning in the domain of Molecular Radiotherapy, has been underlined by the new European council directive (2013/59 Euratom). This represents a great challenge for radionuclide treatments, while they are given more systemically and use different radiotherapeutics available which continue to expand significantly. The new version of PLANET® Dose perfectly meets the current European regulatory requirements to provide a full personalized 3D dosimetry with a patient-oriented approach.
“We are very proud of the new version of PLANET® Dose achieved in collaboration with the CRCT – UMR 1037 Inserm / Toulouse III – Paul Sabatier University and the Institut Jules Bordet. This validation confirms the capability of PLANET® Dose to work with different radionuclides: 90Y-microspheres and 177Lutetium. We are convinced that PLANET® Dose, as the first integrated imaging and dosimetry platform for clinical routine, will help centers in the planning, in vivo control and full traceability of such treatments. It will bring the flexibility for centers to include in the future other potential radionuclides to fight specific cancer cases.” claims Sébastien Vauclin, PLANET Product Manager at DOSIsoft SA.
About DOSIsoft
Founded in 2002, DOSIsoft develops cutting-edge software solutions for Radiation Oncology and Nuclear Medicine. 15 years of innovation and R&D investments have led to world leading software solutions used successfully in over 170 hospital centers in 18 countries around the world. Spin-off of Gustave Roussy and Institut Curie, DOSIsoft constantly innovates in partnership with the major cancer institutes and research centers in the world. www.dosisoft.com
DOSIsoft will participate in the Annual Meeting of EANM from October 13th to 17th, 2018 in Düsseldorf, Germany. Come visit us at booth #41 (Hall4).
